by Dr. Yashashwini Reddy | Aug 8, 2025
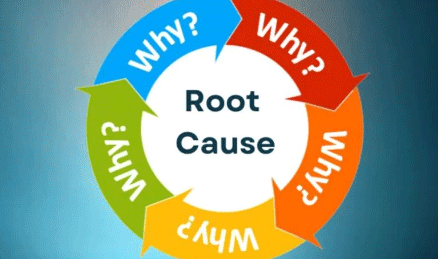
5 Why Tool – Overview The 5 Why Tool is a simple but powerful problem-solving method used to identify the root cause of an issue by repeatedly asking “Why?” until the underlying reason is found. Typically, five iterations of “Why” are enough to reveal the true root...

by Dr. Yashashwini Reddy | May 7, 2025
Standard Operating Procedure (SOP) 1. Objective To establish standardized procedures for reprocessing and reworking pharmaceutical products to ensure that all batches meet predefined specifications and maintain product quality. 2. Scope...

by Dr. Yashashwini Reddy | May 7, 2025
SOP for Failure Investigation in Pharmaceutical Manufacturing Here’s an overview of the Standard Operating Procedure (SOP) for Failure Investigation in pharmaceutical manufacturing, focusing on identifying root causes, implementing corrective and preventive...
by Dr. Yashashwini Reddy | May 5, 2025
Tips for Preventing and Troubleshooting Manufacturing Deviations Manufacturing deviations are any unintended differences or variations from the planned production process or product specifications. These can result in quality issues, production delays, and increased...
by Dr. Yashashwini Reddy | Apr 29, 2025
Analysis of Out of Specification (OOS) Occurrences in Pharmaceutical Products In the pharmaceutical industry, the term Out of Specification (OOS) refers to situations where the results of tests or measurements fall outside the predefined limits or specifications set...







