
by Dr. Yashashwini Reddy | Apr 23, 2025
Standard Operating Procedure (SOP) Here is a Standard Operating Procedure (SOP) for the Preservation of Control Samples of Active Ingredients. This SOP ensures that control samples of active ingredients (API) are preserved and stored in...

by Dr. Yashashwini Reddy | Apr 20, 2025
Standard Operating Procedure (SOP) 1. Purpose: To define the procedure for managing and handling non-conforming products (NCP) in order to identify, segregate, and address quality issues, ensuring that only conforming products are released for use,...

by Dr. Yashashwini Reddy | Apr 20, 2025
Standard Operating Procedure (SOP) 1. Purpose: To define the procedure for monitoring and ensuring product quality during the manufacturing process. 2. Scope: Applies to all manufacturing batches processed in the facility. 3. Responsibility: IPQC...
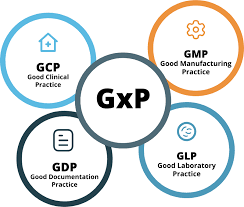
by Dr. Yashashwini Reddy | Apr 19, 2025
1. What are GLP, GDP, and GMP, and how do they differ? This foundational question tests your understanding of the definitions: GLP: Ensures integrity and quality of non-clinical lab studies. GDP: Ensures data integrity and accuracy in documentation. GMP: Ensures...







