
by Dr. Yashashwini Reddy | May 2, 2025
Common Sources of Contamination in Pharmaceutical Manufacturing Contamination in pharmaceutical production can lead to serious consequences, including compromised product quality, patient safety risks, regulatory violations, and financial losses. Contamination can be...

by Dr. Yashashwini Reddy | May 1, 2025
Importance of Calibration in UV/Visible Spectrophotometry Calibration of a UV/Visible spectrophotometer is essential for ensuring the accuracy, reliability, and precision of measurements. Here’s why it is important: Accuracy of Measurements: Calibration ensures that...

by Dr. Yashashwini Reddy | May 1, 2025
Creating a detailed checklist for an audit in Quality Control (QC) is essential for ensuring that processes are evaluated consistently and effectively. Here’s a step-by-step guide to help you create a comprehensive QC audit checklist: 1. Define the Purpose and Scope...

by Dr. Yashashwini Reddy | May 1, 2025
Requirements and Implementation of Continuous Training in Pharmaceuticals Continuous training in the pharmaceutical industry is critical for maintaining high standards of quality, ensuring compliance with regulatory guidelines, and keeping employees updated on the...
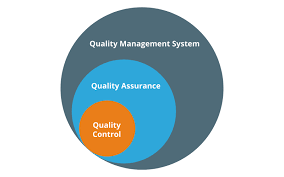
by Dr. Yashashwini Reddy | Apr 29, 2025
Supplier audits have a significant and multifaceted impact on quality assurance in the pharmaceutical sector. Here’s a detailed breakdown of their influence: 1. Ensuring Compliance with Regulatory Standards Supplier audits help ensure that raw material...







