
by Dr. Yashashwini Reddy | Aug 10, 2025
Handling of Out of Calibration (OOC) Instruments and Equipment Definition Out of Calibration (OOC) refers to an instrument or equipment that, during calibration verification, fails to meet the specified acceptance criteria or shows results outside its defined...

by Dr. Yashashwini Reddy | Aug 10, 2025
Why Dissolution Test Apparatus Calibration with Salicylic Acid Tablets was Stopped Historically, salicylic acid tablets were used as calibration standards for dissolution test apparatus (mainly USP Apparatus 1 & 2) to check performance. However, this practice was...
by Dr. Yashashwini Reddy | Aug 10, 2025
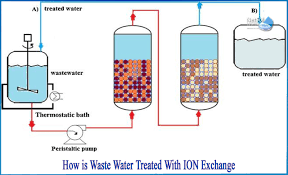
Use of Ion Exchange Resins in Water Purification Systems Ion exchange resins are insoluble, cross-linked polymer beads with functional groups that can exchange specific ions from water, effectively removing unwanted dissolved salts, minerals, or contaminants. They are...
by Dr. Yashashwini Reddy | Aug 9, 2025
Common Causes of Low Quality in Pharmaceuticals Ensuring high-quality pharmaceuticals is crucial to patient safety, regulatory compliance, and brand reputation. Low-quality products can lead to therapeutic failure, adverse reactions, and recalls. The following are...
by Dr. Yashashwini Reddy | Aug 9, 2025
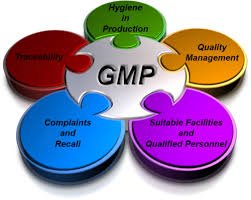
GMP Requirements in Pharmaceuticals Good Manufacturing Practice (GMP) is a regulatory framework ensuring that pharmaceutical products are consistently produced and controlled according to quality standards, minimizing risks to patients. 1. Quality Management System...







