
by Dr. Yashashwini Reddy | Aug 11, 2025
Principle of UV-Vis Spectrophotometry The fundamental principle of UV-Vis spectrophotometry is based on the absorption of ultraviolet (190–400 nm) and visible (400–800 nm) light by molecules. When a molecule absorbs light, electrons are promoted from a lower energy...

by Dr. Yashashwini Reddy | Aug 10, 2025
QC Audit Checklist 1. General Documentation & Records SOPs for all QC activities (latest approved versions, controlled copies). Records of training, competency assessments, and analyst qualification. QC logbooks (instrument usage, calibration, maintenance). Change...
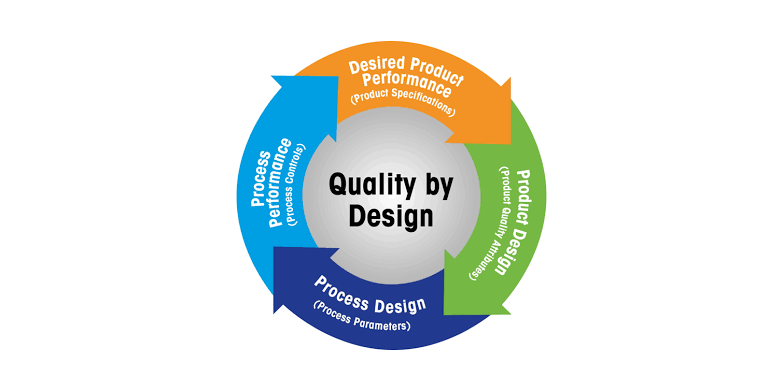
by Dr. Yashashwini Reddy | Aug 10, 2025
Quality by Design (QbD) in Pharmaceuticals Definition:Quality by Design (QbD) is a systematic, risk-based, proactive approach to pharmaceutical development that emphasizes designing and building quality into the product from the very beginning, rather than relying...

by Dr. Yashashwini Reddy | Aug 10, 2025
Requirements and Implementation of Continuous Training in Pharmaceuticals Continuous training in the pharmaceutical industry is a regulatory, quality, and operational necessity to ensure that employees remain competent, compliant, and up-to-date with evolving...

by Dr. Yashashwini Reddy | Aug 10, 2025
Calibration Frequency of Instruments and Other Measuring Devices In a pharmaceutical GMP environment, calibration frequency is determined based on regulatory guidelines, manufacturer recommendations, risk assessment, and historical performance of the instrument....







