by Dr. Yashashwini Reddy | Sep 13, 2025
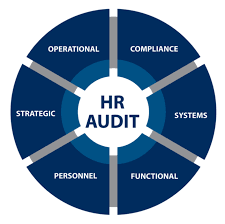
HR Audit Checklist 1. Recruitment & Selection Existence of recruitment policies and SOPs. Proper manpower requisition approvals before hiring. Background verification and reference checks of employees. Job descriptions and competency requirements documented....

by Dr. Yashashwini Reddy | Sep 11, 2025
📌 Data Falsification in the Pharmaceutical Industry Definition:Data falsification is the intentional alteration, manipulation, or fabrication of data in order to misrepresent results and meet regulatory, quality, or business expectations. It is one of the most serious...

by Dr. Yashashwini Reddy | Sep 11, 2025
📌 Regulatory Compliance in Pharmaceuticals: 8 Common Mistakes and How to Avoid Them Definition:Regulatory compliance in pharmaceuticals means adhering to laws, regulations, and guidelines (e.g., US FDA, EMA, WHO, MHRA, CDSCO) to ensure medicines are safe, effective,...

by Dr. Yashashwini Reddy | Sep 11, 2025
📌 Contamination Issues in Pharmaceutical Production and Their Prevention Definition:Contamination in pharmaceuticals refers to the undesired introduction of chemical, microbial, or physical material into drug products during manufacturing, packaging, storage, or...

by Dr. Yashashwini Reddy | Sep 11, 2025
📌 Computerized System Validation (CSV) in Pharmaceuticals Definition:Computerized System Validation (CSV) is a documented process of ensuring that computerized systems used in GxP (Good Practice) environments consistently function as intended and comply with...







