
by Dr. Yashashwini Reddy | Oct 9, 2025
Continuous Manufacturing (CM) is an advanced pharmaceutical production approach where input materials are continuously fed into the system and the processed output (finished product) is continuously removed. Unlike traditional batch manufacturing, which occurs in...
by Dr. Yashashwini Reddy | Oct 8, 2025
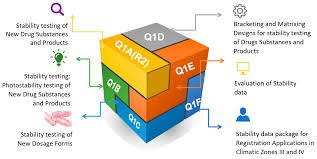
📘 ICH Q1: Stability Testing 🔹 Purpose The ICH Q1 series provides guidance on how to design stability studies to ensure that drug substances and drug products maintain their quality, safety, and efficacy throughout their shelf life under various environmental...

by Dr. Yashashwini Reddy | Oct 8, 2025
🧭 Quality Guidelines – Overview Quality Guidelines are internationally harmonized standards developed mainly by the International Council for Harmonisation (ICH) and other regulatory authorities (like FDA, EMA, WHO, CDSCO).They help ensure that pharmaceutical products...

by Dr. Yashashwini Reddy | Sep 29, 2025
Formats of Pharmaceutical Audits On-Site Audit (Physical Audit) Conducted at the manufacturing site, laboratory, warehouse, or supplier facility. Provides direct observation of processes, equipment, and personnel. Most common format for GMP compliance checks. Remote...

by Dr. Yashashwini Reddy | Sep 29, 2025
In the pharmaceutical industry, quality audits are critical for ensuring compliance with Good Manufacturing Practices (GMP), regulatory guidelines, and internal quality standards. The main types of audits include: Types of Quality Audits in Pharma Internal Audit...







