
by Dr. Yashashwini Reddy | Sep 29, 2025
Aspect Audit Inspection Definition A systematic, independent review to assess compliance with internal or external standards (e.g., GMP). A formal review by a regulatory authority (e.g., FDA, EMA) to ensure compliance with laws and regulations. Conducted By Internal...

by Dr. Yashashwini Reddy | Sep 24, 2025
Case Study: Valsartan Recall (2018) Background Valsartan is an angiotensin II receptor blocker (ARB) widely prescribed for hypertension and heart failure. In July 2018, several regulatory agencies, including the USFDA, EMA, and Health Canada, announced recalls of...

by Dr. Yashashwini Reddy | Sep 23, 2025
Case Study: Supply Chain Disruption – API Shortage Background A U.S.-based pharmaceutical company manufacturing essential cardiovascular and anti-diabetic medicines faced a sudden API (Active Pharmaceutical Ingredient) shortage in 2024. The primary API supplier was...
by Dr. Yashashwini Reddy | Sep 23, 2025
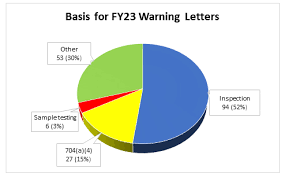
📌 Case Study: Data Integrity Failures – FDA Warning Letters 1. Background In recent years, several pharmaceutical companies (especially in India and China) have faced FDA Warning Letters and import alerts because of data integrity violations. These cases highlight the...
by Dr. Yashashwini Reddy | Sep 23, 2025
🔹 For Patients Access to New Treatments Early Patients may gain access to investigational drugs, biologics, or devices before they’re widely available. This can be especially important when current treatments are limited or ineffective. Closer Medical Monitoring...







