by Dr. Yashashwini Reddy | Apr 19, 2025
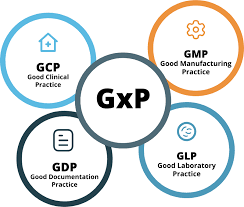
1. What are GLP, GDP, and GMP, and how do they differ? This foundational question tests your understanding of the definitions: GLP: Ensures integrity and quality of non-clinical lab studies. GDP: Ensures data integrity and accuracy in documentation. GMP: Ensures...

by Dr. Yashashwini Reddy | Apr 10, 2025
To embark on a career in Regulatory Affairs, it is essential to obtain a bachelor’s degree in a life sciences discipline, acquire relevant experience through internships or entry-level positions, and contemplate pursuing advanced degrees or certifications to...

by Dr. Yashashwini Reddy | Apr 10, 2025
A typical career trajectory in Regulatory Affairs may include the following positions: – Regulatory Officer/Executive/Associate – Senior Associate – Manager – Senior Manager – Director – Senior Director – Head of Department...

by Dr. Yashashwini Reddy | Apr 5, 2025
The career paths in Pharmacovigilance or Drug Safety are both – knowledge and skill-based. One’s educational background in the field of medicine, be it – a physician, a dentist, a pharmacology student (and so on) – along with the skill sets acquired...
by Dr. Yashashwini Reddy | Mar 30, 2025
Module 3: Quality (CMC) in Regulatory Dossier Submission In pharmaceutical regulatory submissions, Module 3 of the Common Technical Document (CTD) plays a critical role in ensuring drug quality, safety, and efficacy. This module, also known as the Quality Module,...






