by Dr. Yashashwini Reddy | Oct 9, 2025
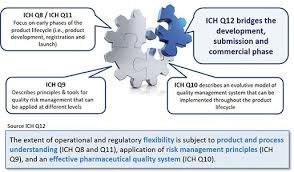
Lifecycle Management (LCM) in the pharmaceutical industry refers to the systematic approach of managing a product from its development phase through commercialization, post-approval changes, and discontinuation. It ensures continuous product quality, compliance, and...
by Dr. Yashashwini Reddy | Oct 8, 2025
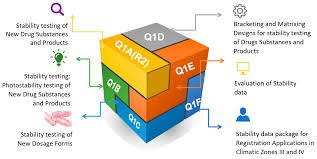
📘 ICH Q1: Stability Testing 🔹 Purpose The ICH Q1 series provides guidance on how to design stability studies to ensure that drug substances and drug products maintain their quality, safety, and efficacy throughout their shelf life under various environmental...





