
by Dr. Yashashwini Reddy | Sep 15, 2025
FDA Forms Generally Used in Pharmaceutical Inspection 1. Form FDA 482 – Notice of Inspection Issued at the start of an inspection. Notifies the company that FDA investigators are officially beginning the inspection under the authority of the FD&C Act (Section...
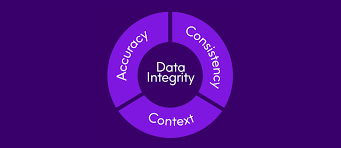
by Dr. Yashashwini Reddy | Sep 9, 2025
🔐 Why Data Integrity is More Important Than Ever? 1. Patient Safety at the Core Medicines are only as safe as the data proving their quality. Any falsified, incomplete, or inaccurate record may lead to unsafe products reaching patients. Strong data integrity ensures...

by Dr. Yashashwini Reddy | Sep 9, 2025
🏭 How FDA Inspections are Conducted in Manufacturing Facilities 1. Pre-Inspection Phase FDA identifies facilities to inspect based on: Risk-based selection (product type, compliance history, criticality, recalls, complaints). New drug approval or pre-approval...

by Dr. Yashashwini Reddy | Sep 9, 2025
🧫 Typical Microbiology Concerns in an FDA Inspection 1. Environmental Monitoring (EM) Deficiencies Inadequate EM program for cleanrooms and controlled areas. Failure to establish alert/action limits based on historical data. Poor trending and lack of investigation of...

by Dr. Yashashwini Reddy | Aug 18, 2025
Top 5 Tips for a Successful FDA Inspection: ✅ Top 5 Tips Be Inspection-Ready Always – Maintain compliance at all times, not just when expecting an inspection. Ensure documentation, training records, SOPs, and facilities are always up-to-date. Ensure Documentation...







